Latest News

Latest News

Latest News
Everfront Biotech to Present Updated Cerebraca® Wafer Clinical Trial Results at the SNO 2025 Annual Meeting
Date: November 17, 2025
Everfront Biotech has been selected for a Poster Presentation at the conference. The company will unveil the latest updated results from its Phase I/IIa clinical trial of Cerebraca® Wafer for the treatment of Recurrent Glioblastoma (GBM), and will also outline its plans for the upcoming Phase IIb/III clinical trial. Cerebraca® Wafer is one of Everfront Biotech’s core products, focused on addressing multiple challenges in the treatment of malignant brain tumors…
Everfront Biotech’s Novel Brain Tumor Implant Drug Cerebraca® Wafer Gained International Recognition, to Present Global Pivotal Clinical Trial Plans at 2025 CNS Annual Meeting in the U.S.
Date: October 14, 2025
The results of the Phase I/IIa clinical trial for Everfront Biotech’s innovative implantable drug for the local treatment of Glioblastoma (GBM), Cerebraca® Wafer, made their international debut at the Congress of Neurological Surgeons (CNS) Annual Meeting, held in Los Angeles, California, USA, from October 11 to 15, 2025. Dr. Jen-Wei Liu, Chief Science Officer of Everfront Biotech, noted that Dr. Clark Chen, the company’s Scientific Advisory Committee member and a renowned neurosurgery expert in the US, delivered an oral presentation at the CNS Annual Meeting. The event attracted hundreds of experts and scholars, who attended the session and engaged in enthusiastic discussions afterward.
Everfront Biotech to Unveil Breakthrough Clinical Trial Results for Cerebraca® Wafer in Brain Cancer Treatment at 2024 Taiwan Healthcare+ Expo
Date: November 28, 2024
Everfront Biotech has been invited to participate in the expert Lecture hosted by the Hualien Tzu Chi Medical Center at the 2024 Taiwan Healthcare+ EXPO. Dr. Jen-Wei Liu, Chief Scientific Officer at Everfront Biotech, will deliver a groundbreaking presentation titled “Redefining Malignant Brain Tumor Treatment: Current Developments and Future Prospects of the Cerebraca® Wafer.” This lecture will unveil the results of the recently completed Phase I/IIa clinical trials and provide insights into the upcoming multinational, multicenter clinical trial plans, as well as the company’s future prospects.
Taiwan’s Biotech and Pharma Delegation: Innovations Take Center Stage at BIO 2024
Date: June 21, 2024
Taiwan’s biotechnology industry is making a significant impact at the 2024 BIO International Convention in San Diego. Organized by the Biotechnology and Pharmaceutical Industries Promotion Office (BPIPO) under the Ministry of Economic Affairs, the Taiwan delegation includes representatives from various government bodies, research institutions, and over 60 delegates.
Everfront Biotech showcases its latest drug development achievements, particularly the innovative treatment for glioblastoma, Cerebraca® Wafer. This FDA-designated orphan drug offers localized, sustained drug delivery post-surgery, overcoming the blood-brain barrier. Clinical trials indicate a median survival period significantly higher than existing treatments. Everfront’s focus on unmet medical needs and orphan drug development positions it as a key player in pioneering cancer therapies.
Dr. Shinn-Zong Lin Shares Clinical Insights on Cerebraca® Wafer at the Cross-Strait Traditional Chinese Medicine Development Symposium
Date: June 19, 2024
Dr. Shinn-Zong Lin, Director of Hualien Tzu Chi Hospital, attended the Cross-Strait Traditional Chinese Medicine Development and Cooperation Symposium held in Xiamen on June 14. In addition to delivering a speech at the opening ceremony, Dr. Lin, along with Vice President Tsung-Jung Ho, shared the results of cross-team collaboration in integrating traditional Chinese and Western medicine to treat cancer, rare diseases, and brain injuries such as stroke during the keynote forum. Dr. Lin also shared his experiences and results from the Phase IIa clinical trial of the new drug Cerebraca® Wafer, aimed at combating malignant glioblastoma.
Everfront Biotech at BIO 2024: Showcasing Clinical Achievements alongside 47 Global Startups
Date: June 8, 2024
Everfront Biotech will compete with 47 innovative biotech companies from around the world at the BIO 2024 Start-Up Stadium on the afternoon of June 4. Dr. Jui-Hao Lee, Director of the Clinical Medicine Department, will represent Everfront Biotech and present the latest results from the Phase IIa clinical trial of Cerebraca® Wafer to the attendees. Compared to the current standard therapy, which has a median overall survival of 6-9 months, patients receiving the highest dose of 6 Cerebraca® Wafers achieved a median survival of 26.2 months. Furthermore, these patients were able to be discharged earlier and enjoy a better quality of life, representing a significant breakthrough for both patients’ families and the broader healthcare system.
Everfront Biotech at BIO 2024: Competing in the Taiwan Smart Health GALA with 22 Startups
Date: June 3, 2024
On June 3 (U.S. time), the Taiwan Smart Health GALA, organized by the National Science and Technology Council (NSTC) and co-organized by ITRI and StartupIsland, will be grandly unveiled during the BIO 2024 International Convention. The event features distinguished speakers such as Peter Kurz, Chief Strategy Officer of Quantum International Corp (QIC), and Dr. Jau-Hua Lin, CEO of PharmaEssentia, who will share Taiwan’s formidable capabilities in biotech and pharmaceutical R&D.
Following the expert keynote speeches, Everfront Biotech, along with 22 other leading Taiwanese biotech companies and startups, will compete on the same stage. Dr. Jui-Hao Lee, Director of the Clinical Medicine Department, will present Everfront Biotech’s latest clinical trial results and breakthroughs in a two-minute presentation to the attendees.
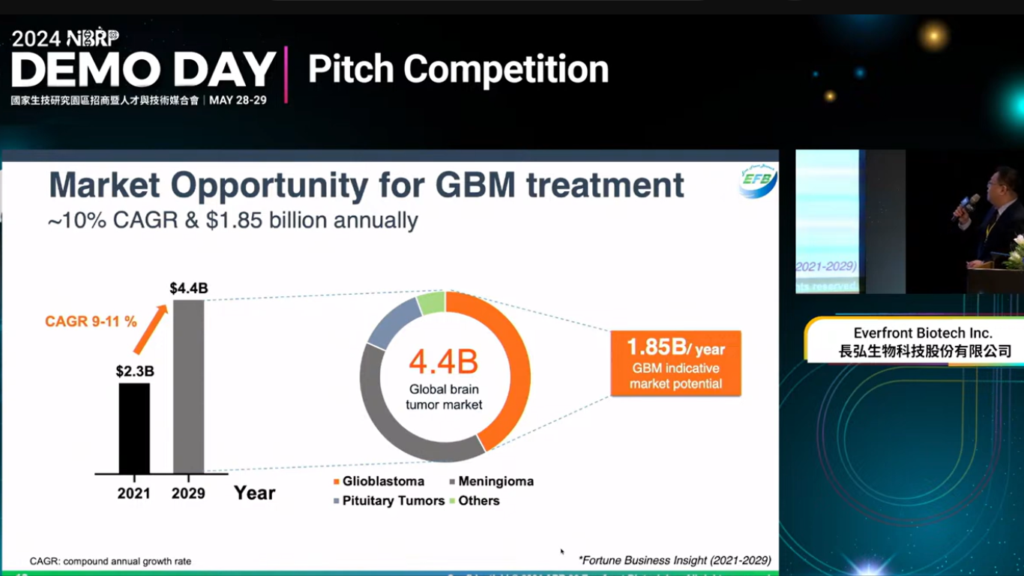
Everfront Biotech Participates in the 2024 National Biotechnology Research Park Demo Day Startup Pitch Competition
Date: May 31, 2024
On May 29, an exciting pitch competition will be held, featuring top talents from the domestic biotech industry, government, and academia. This event will showcase innovative research and development achievements, with startup investors and industry experts serving as judges, providing comprehensive feedback to participants, and increasing opportunities for collaboration and investment partnerships. Representing Everfront Biotech, Dr. Jen-Wei Liu, Chief Scientific Officer, will present on the topic: “Integrated Therapeutic Solutions: Enhancing Tumor Treatment with the Combination of Localized Release from Cerebraca® Wafer and Systemic Delivery via EF-031 Softgel Capsules.”
Five Teams Compete for World Championship as NSTC Leads 16 Startups to North America’s Premier Biotech Event
Date: May 31, 2024
The global biotech event, the North American Biotech Exhibition, will take place from June 3 to 6. The NSTC will lead 16 Taiwanese startups to participate, including Everfront Biotech, Suntec Medical, Trust Bio-sonics, AcroCyte Therapeutics, and CytoArm. These five teams have successfully advanced to the final round of the Start-Up Stadium competition, where they will compete for the world championship against 42 other teams from around the globe. The NSTC stated that Everfront, Suntec, and Aether were selected for their revolutionary drug delivery technologies, which have the potential to overcome the limitations of traditional drug delivery methods, such as ineffective treatment and damage to other tissues. Their innovations offer advantages such as precise cancer medication delivery and immune system activation.
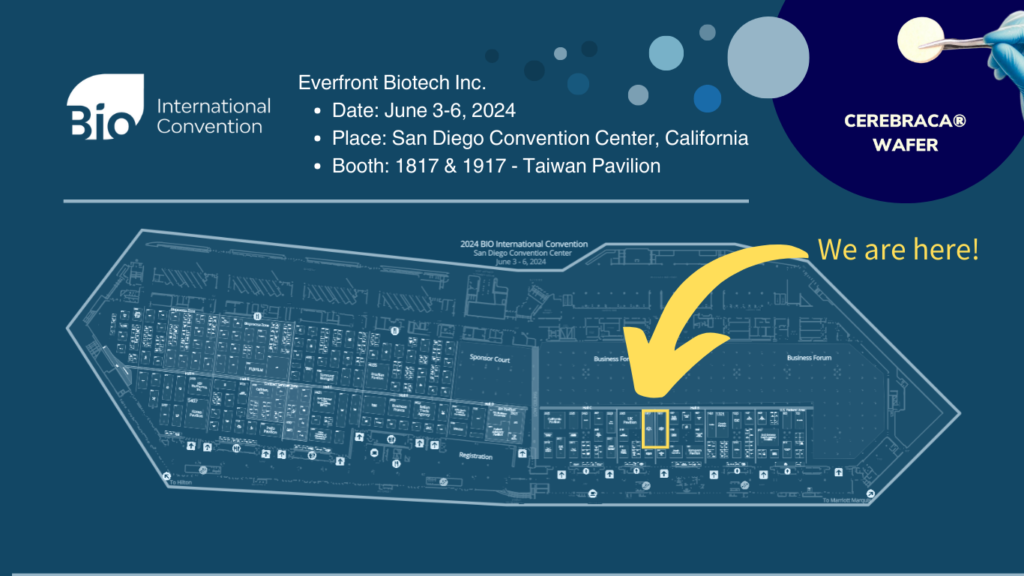
Everfront Biotech will attend the BIO 2024 International Convention to showcase the latest achievements in new drug development
Date: April 15, 2024
Everfront Biotech will attend the BIO 2024 International Convention, held in San Diego, United States, from June 3rd to June 6th, 2024. This event is recognized as the largest international exhibition and forum in the global biotechnology industry, representing the pinnacle of the biotech world annually. We will join the Taiwan Biotech Delegation and exhibit at Booth 1817 & 1917 within the Taiwan Pavilion, alongside 54 other elite domestic companies. We will showcase Everfront Biotech’s recent clinical achievements, aiming to align Taiwan’s biotech capabilities with the international community and seize more international collaboration opportunities. We look forward to your visit.
Everfront Biotech Team Receives 2023 National Innovation Award
Date: Janurary 18, 2024
Everfront Biotech Inc. was honored with the 20th National Innovation Award on December 25, 2023, recognizing our outstanding achievements in the field of combating glioblastoma multiforme (GBM) with Cerebraca® Wafer. The clinical trial program successfully concluded the Phase IIa clinical trial at Hualien Tzu Chi Medical Center, Tri-Service General Hospital, and Taichung Veterans General Hospital in April 2023. In June of the same year, it passed the review of the Fast Track program by the Ministry of Economic Affairs. We anticipate completing the clinical trial report in 2024 and subsequently engaging in in-depth communication with the U.S. FDA to prepare for the next pivotal clinical trial phase.
Highlights of Everfront Biotech at the Taiwan Healthcare+ EXPO 2023
Date: December 2, 2023
Everfront Biotech was invited to collaborate with Hualien Tzu Chi Medical Center for the 2023 Taiwan Medical Technology Exhibition. A specialist lecture, themed “The Cerebraca® Wafer Journey to the Global Stage” was held on December 2nd at 1:00 PM. The event attracted numerous enthusiastic attendees, and post-event discussions involved many experts and scholars. The expectation is to swiftly progress into the Phase IIb stage with multiple international centers and successfully conclude patient enrollment.
New Targeted Drug Cerebraca® Wafer: Dawn of Treatment for Malignant Brain Tumors
Date: November 21, 2023
The current treatment efficacy for recurrent malignant gliomas is limited, demanding urgent exploration of new therapeutic approaches. On December 2nd at 1:00 PM, Dr. Shinn-Zong Lin, the Superintendent of Tzu Chi Hospital, along with Director Jen-wei Liu and Director Yuan-Sheng Li from Everfront Biotech, jointly presented the latest developments in the new drug for malignant brain tumors, “The Cerebraca® Wafer Journey to the Global Stage.” at the Hualien Tzu Chi Hospital booth (Booth N802) on the 4th floor of Hall 1 at the Healthcare+ EXPO Taiwan 2023 in Taipei Nangang Exhibition Center.
Everfront Participate Healthcare+ EXPO Taiwan 2023
Date: November 9, 2023
Everfront Biotech will participate in Healthcare+ EXPO Taiwan 2023, a not-to-be-missed event in the Asia-Pacific medical biotechnology arena. It’s also a showcase of the domestic biomedicine industry’s strength on the global stage. We are honored to be invited to exhibit alongside Hualien Tzu Chi Medical Center (Booth N802, 4F) during this event. We will present the clinical progress of our anti-brain cancer drug and our research achievements in treating neurodegenerative diseases. We look forward to your visit.
Integrative Treatment of Traditional Chinese and Western Medicine: Active Small Molecule Drugs Derived from Angelica Sinensis Aid in Treating Brain Cancer and Alzheimer's Disease
Date: August 14, 2023
Dr. Shinn-Zong Lin, the Superintendent, delivered a presentation on the topic “Integrative Treatment of Traditional Chinese and Western Medicine – Neurological Disorders as an Example: Tumor Elimination and Anti-Aging.” He shared insights about leading a team, which included Vice Research and Development Director Horng-Jyh Harn from Tzu Chi Innovation Research Center and Distinguished Professor Tzyy-Wen Chiou from National Donghua University, in discovering the therapeutic potential of an extract from Angelica Sinensis in cancer treatment. They identified the active small molecule compound BP and developed targeted small molecule anticancer drugs for the treatment of malignant brain gliomas. Furthermore, they combined BP with CPPSA copolymer to create the Cerebraca® wafer, a sustained-release wafer, which delivers the targeted small molecule drug BP to brain tumor lesions. This innovative approach has gained patents in countries including the United States, Mainland China, Japan, the European Union, and Taiwan.
Highlights from Everfront Biotech at BIO ASIA-Taiwan 2023
Date: July 27, 2023
Everfront Biotech has been invited to participate in the National Science Council’s Startup Zone, where it showcased its latest developments. On the 27th, the company took part in the Precision Marketing (Elevator Pitch) organized by the National Science Council, followed by the Company Presentations on the 28th. The event was filled with enthusiasm and provided an excellent platform for the public to gain deeper insights into Everfront Biotech’s endeavors.
Everfront Participate BIO ASIA-Taiwan 2023 Exihibition
Date: July 4, 2023
Everfront Biotech will participate in BIO ASIA–Taiwan 2023, the largest biotechnology industry event in Asia, from July 27 to July 30, 2023. This international biotech extravaganza is not to be missed. We are delighted to announce that our booth (NSTC INNO ZONE, 4F, N814) has been selected in the Nangang Exhibition Hall. We will showcase our recent achievements in new drug development. We look forward to your visit and participation.
Everfront Biotech Inc. Receives "Investor's Choice Award" at the 2023 NBRP Demo Day Investment Matchmaking Competition
Date: May 5, 2023
Everfront Biotech’s theme for this year’s competition was “Revolutionizing Brain Cancer Treatment with Cerebraca® Wafer,” which stood out and won the “Investor’s Favorite Award” at the NBRP Demo Day investment matchmaking competition. In a limited amount of time, Dr. Lee introduced Everfront Biotech’s research and development capabilities, the pharmacological mechanism, precise release characteristics, preliminary results from phase I clinical trials, and compared them with current treatment methods, all of which sparked a high level of interest and enthusiasm among the attending investors. This presentation led to Everfront Biotech Inc. being recognized by the investors and awarded the prize, which demonstrated their strong willingness to invest in Cerebraca® Wafer…
Our Team Is Honored with the National Innovation Award – Clinical Innovation for Treating Recurrent Malignant Brain Tumors
Date: December 6, 2022
Six years ago, commissioned by Everfront Biotech Inc., the team from Hualien Tzu Chi Hospital, in collaboration with the neurosurgery team from Tri-Service General Hospital, carried out clinical trials for the development of the new drug Cerebraca® wafer for the treatment of malignant brain tumors…
Everfront Biotech's Investigational product, Cerebraca® Wafer, has achieved significant progress in Phase I clinical trials for the treatment of malignant brain tumors, and has been invited to speak at the Asian Congress of Neurological Surgeons (ACNS).
Date: June 9, 2022
Hualien Tzu Chi Hospital collaborated with the neurosurgery team from Tri-Service General Hospital to conduct clinical trials for the development of a new drug, Cerebraca® Wafer, for the treatment of malignant brain tumors, under the leadership of the Superintendent, Dr. Shinn-Zong. Dr. Lin was invited by the President of the Asian Congress of Neurological Surgeons, Professor Yoko Kato, to share the results of the phase I clinical trial via video conference on June 8th at 8:30 PM.
Everfront Biotech's Cerebraca® Wafer has Completed the Phase I - Safety Evaluation for the Treatment of Glioblastoma Multiforme, and has Initiated the Phase IIa - Efficacy Evaluation
Date: April 29, 2022
Everfront Biotech Inc., a company focused on developing new drugs for over 20 years, specializing in cancer, neurodegenerative, and rare diseases, has developed a small molecule targeted therapy for glioblastoma multiforme called Cerebraca® Wafer. The drug will complete phase II trials at Hualien Tzu Chi Hospital, Taipei Tri-Service General Hospital, and Taichung Veterans General Hospital, and is preparing to publish its findings in the renowned international journal, Cancers. This breakthrough from a local Taiwanese research team has not only energized the medical community, but also prompted the company to seek international collaborations through an initial public offering.
The Research Team for Developing a New Drug, EF-009, for the Treatment of Pancreatic Cancer Won the National Innovation Award
Date: November 16, 2021
The preclinical development of EF-009 has been completed, and the process meets the international standards of PIC/S GMP for the production of active pharmaceutical ingredient, excipient, and drug product – Cerebraca® wafer. Results from toxicity studies, safety pharmacology studies, genotoxicity studies, and pharmacokinetic studies have preliminary confirmed its safety and drug metabolism characteristics.
This new drug has been transferred to Everfront Biotech Inc. and has received Investigational New Drug (IND) approval from the USFDA and the Taiwan FDA. It will soon enter Phase I/IIa trial to test its safety and efficacy in humans, with the hope of helping pancreatic cancer patients in the future.
Targeted Small Molecules can Enhance Treatment for Glioblastoma Multiforme, winning the National Innovation Award
Date: December 7, 2019
Superintendent Shinn-Zong Lin led the new drug innovation and development team to discover the targeted small molecule drug EF-001, which is used to inhibit the cancer immune checkpoint PD-L1, activate immune T cells, and enhance the immunotherapy effect of glioblastoma multiforme. This is a major breakthrough in global immunotherapy research for glioblastoma multiforme and has been recognized by the National Innovation Award with the “Academic Research and Innovation Award” …
"2019 Taipei Biotech Award": Ko Wen-Je Claims Biotech to be the Most Potential Industries
Date: July 26, 2019
“The 2019 Taipei Biotech Awards were divided into three categories: the New Technology Award, the International Breakthrough Award, and the Technology Transfer and Collaboration Award, with a total of 15 finalists. Action Gene took home the gold award in the New Technology category, receiving the highest prize of 1 million NTD. Taiwan Biotech Co. (6589) and Everfront Biotech Inc. were awarded the Excellence Award in the same category.”
Everfront Biotech Inc. and Buddhist Tzu Chi Hospital Jointly Develop New Drugs
Date: March 24, 2018
Everfront Biotech Inc. successfully completed its Phase I trial application at Buddhist Tzu Chi General Hospital in Hualien, Taiwan and Tri-Service General Hospital in Taipei, Taiwan. The clinical trial was initialed on December 25th, 2017, with the Superintendent, Dr. Shinn-Zong Lin and Dr. Tsung-Lang Chiu from Tzu Chi General Hospital and Dr. Sin-I Ma, Dr. Yuan-Hao Chen, Dr. Tung-Yuan Hong, and Dr. Wei-Hsiu Liu from Tri-Service General Hospital as the principal investigators. The Phase I human trial study aimed to treat patients with high grade recurrent glioblastoma multiforme using brain-targeted drugs implanted into the brain. With the completion of this trial, Everfront Biotech Inc. has made significant progress towards its goal of developing drugs to treat malignant brain tumors.
Collaboration between Lotus Pharma, Tzu Chi Hospital and Everfront Biotech for Clinical Trials of New Drugs for Glioblastoma Multiforme in June
Date: May 22, 2017
Five years ago, Everfront Biotech Inc. acquired the key technology from Hualien Tzu Chi Hospital and National Dong Hwa University to develop Cerebraca® Wafer, a targeted anti-cancer drug for the treatment of glioblastoma multiforme. Cerebraca® Wafer slowly releases and diffuses into the surrounding brain tissue, eliminating residual tumor cells that were not completely removed during surgery. By inhibiting the DNA repair gene MGMT, which causes resistance to Temozolomide, malignant cells that were resistant to Temozolomide can be killed again. Currently, Cerebraca® Wafer has been granted patents in Taiwan, China, the United States, Japan, and the European Union. Since August 2016, it has obtained clinical trial approval from the US Food and Drug Administration (FDA) and Taiwan Food and Drug Administration (TFDA). In January of this year, Everfront Biotech Inc. signed a contract with Hualien Tzu Chi Hospital to conduct Phase I trial….
Cerebraca® wafer, a Locally Developed Targeted Drug, Brings New Hopes for the Treatment of Glioblastoma Multiforme. Everfront Biotech and Tzu Chi Hospital Have Signed a Contract to Collaborate and Embark on a New Journey
Date: Janurary 26, 2017
On January 26th, Everfront Biotech Inc. signed a letter of intent with Hualien Tzu Chi Hospital to commission the hospital to conduct the Phase I/IIa trial of the targeted new drug Cerebraca® Wafer for the treatment of glioblastoma multiforme. The clinical trial aims to test the safety and effectiveness of the new drug in humans and hopes to help patients with malignant brain tumors in the future.
At 10:00 am on January 26th, Mrs. Pei-Wen Chou, chairman of Everfront Biotech Inc., and Dr. Shinn-Zong Lin, Superintendent of Hualien Tzu Chi Hospital, signed the letter of intent at the hospital.