Cerebraca® wafer, a Locally Developed Targeted Drug, Brings New Hopes for the Treatment of Glioblastoma Multiforme. Everfront Biotech and Tzu Chi Hospital Have Signed a Contract to Collaborate and Embark on a New Journey
Date: Janurary 26, 2017

Everfront Biotech Inc. and Hualien Tzu Chi Hospital signed a memorandum of understanding for new drug development on January 26th. Hualien Tzu Chi Hospital is commissioned to conduct a Phase I/IIa clinical trial of the targeted drug Cerebraca® Wafer for the treatment of glioblastoma multiforme.
On January 26, Everfront Biotech Inc. and Hualien Tzu Chi Hospital signed a letter of intent for a new drug development collaboration, and commissioned Hualien Tzu Chi Hospital to carry out “Phase I/IIa clinical trial of Cerebraca® wafer targeted therapy for glioblastoma multiforme” to test the safety and efficacy of the new drug in humans, with the hope of helping malignant brain tumor patients in the future. The signing ceremony was held at the Cooperation Building of Hualien Tzu Chi Hospital at 10 am, attended by the Everfront Biotech Inc. new drug development team, Hualien Tzu Chi Hospital research team, and Tzu Chi medical volunteers.
Malignant tumors continue to be one of the top ten leading causes of death among Taiwanese, with an estimated 20,000 new brain tumor cases diagnosed each year worldwide. According to statistics from the American Cancer Society (ACS) and the Central Brain Tumor Registry of the United States (CBTRUS), there are about 400 new cases of malignant glioma in Taiwan each year. Glioblastoma multiforme (GBM) is a particularly malignant brain tumor that can grow up to sixteen times its original size within a month. It is a rapidly worsening primary brain tumor, and the recurrence rate after surgery is also very high. Patients diagnosed with grade IV GBM have an average survival time of only 12 to 18 months. The spread of malignant brain tumors is rapid and difficult to eradicate. Once diagnosed, it is usually in the late stage, and the average life expectancy of patients is only about one year, with a five-year survival rate of only 3.4%. Current medical treatments for malignant brain tumors, including surgical treatment, radiation therapy, and chemotherapy, have very limited efficacy.
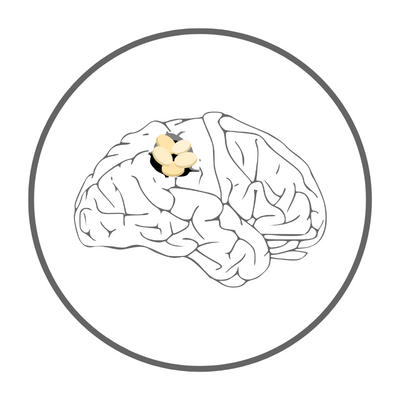
Five years ago, Everfront Biotech’s new drug development team obtained key technology from Hualien Tzu Chi Hospital and National Dong Hwa University through technology transfer. They used EF-API-001 to develop targeted anticancer drugs and made Cerebraca® Wafer from biodegradable biomedical material p(CPP-SA). The targeted genes are Axl/EZH2/SOX2, telomerase, and O6-methylguanine-DNA-methyltransferase. The targeted anticancer drug slowly releases and diffuses into the surrounding brain tissue, removing tumor cells that were not removed during surgery.
The safety and efficacy of Cerebraca® Wafer have been verified in various animal models. For example, in an experiment using rats, the drug was released into the brain tumor area using Cerebraca® Wafer, and the average survival time of the animals was extended by 2.5 times compared to the group that did not receive treatment. Unlike other drugs, the targeted anticancer drug can specifically target tumor cells, reduce the side effects on healthy cells, and enhance the effectiveness of the drug.

Chairman Pei-Wen Chou of Everfront Biotech Inc. (left) signs a contract with Superintendent Shinn-Zong Lin of Hualien Tzu Chi Hospital (right).

“Everfront Biotech’s R&D Director, Dr. Jen-Wei Liu (left), shares an introduction to a new drug.”