Highlights from Everfront Biotech at BIO ASIA-Taiwan 2023
Date: July 27, 2023

Everfront Biotech participated in BIO ASIA-Taiwan 2023, the largest biotechnology exhibition in Asia. We were selected to exhibit in the INNO ZONE of the National Science Council (4F, N814)
The highly anticipated 2023 BIO Asia-Taiwan, jointly organized by Taiwan Bio Industry Development Association and EXPOZAR International Corp., commenced with grandeur on July 27th at Taipei Nangang Exhibition Center, Hall 1. The event, spanning four days, will run until July 30th. With the formal lifting of pandemic restrictions, this year’s exhibition has broken previous records, boasting 14 pavilions, bringing together 800 leading companies from 19 countries, occupying 2,000 booths. Notably surpassing pre-pandemic scales, it has become the largest biotech exhibition in Asia and one of the top three global biotech events, showcasing the explosive growth of Taiwan’s biotech industry.
【Three Key Points in 2023: Record-breaking Scale, Enhanced International Participation, Showcasing Asian Features】
Lee Chung-Hsi, the Chairman of BIO Asia-Taiwan, stated during the pre-event press conference, “The theme of this year’s BIO Asia-Taiwan is ‘Embracing Asian Dynamics,’ encompassing Innovation momentum, Market dynamics, and Investment momentum. These three dynamics hold distinctive features and countless collaboration opportunities in the Asian market. We hope that international attendees can embrace the energy of Taiwan and Asia through this grand event.”
The three main highlights of this year’s BIO Asia-Taiwan are the record-breaking scale, enhanced international participation, and showcasing Asian features. In terms of scale, the exhibition features an unprecedented 2,000 booths, making it the largest biotech expo in Asia. In terms of international participation, there are 19 participating countries, with the USA, UK, Australia, Switzerland, Belgium, Italy, Poland, India, Malaysia, Austria forming 10 national pavilions. Additionally, companies from Japan, China, South Korea, Singapore, Hong Kong, Sweden, Germany, France, Denmark, and other countries are exhibiting. Regional cooperative meetings among multiple countries and in-depth interactions with exhibitors are also taking place. Prominent domestic and international companies such as Pfizer, JCY Biotech, TAIKANG Biotechnology, KYUK, BIOVIA, Biotest, IQVIA, CYTIVA, YUHUA Pharmaceuticals, KoDing, Takeda, and more are present at the event. Furthermore, 11 government-themed pavilions from organizations such as National Science Council, Academia Sinica, Council of Agriculture, National Health Research Institutes, Ministry of Economic Affairs, Department of Chinese Medicine, Industrial Technology Research Institute, along with 9 university research institutions from National Taiwan University, National Cheng Kung University, China Medical University, Taipei Medical University, National Tsing Hua University, will collectively showcase the innovative progress in Asia and Taiwan in areas such as new drugs, therapies, precision health, and smart healthcare, fully demonstrating Asian characteristics.
Source: https://expo.bioasiataiwan.com/newsDetail.asp?serno=4356
Everfront Biotech has been invited to participate in the National Science Council’s Startup Zone, where it showcased its latest developments. On the 27th, the company took part in the Precision Marketing (Elevator Pitch) organized by the National Science Council, followed by the Company Presentations on the 28th. The event was filled with enthusiasm and provided an excellent platform for the public to gain deeper insights into Everfront Biotech’s endeavors. (Please refer to the schedule in the attached image for the speaking times).
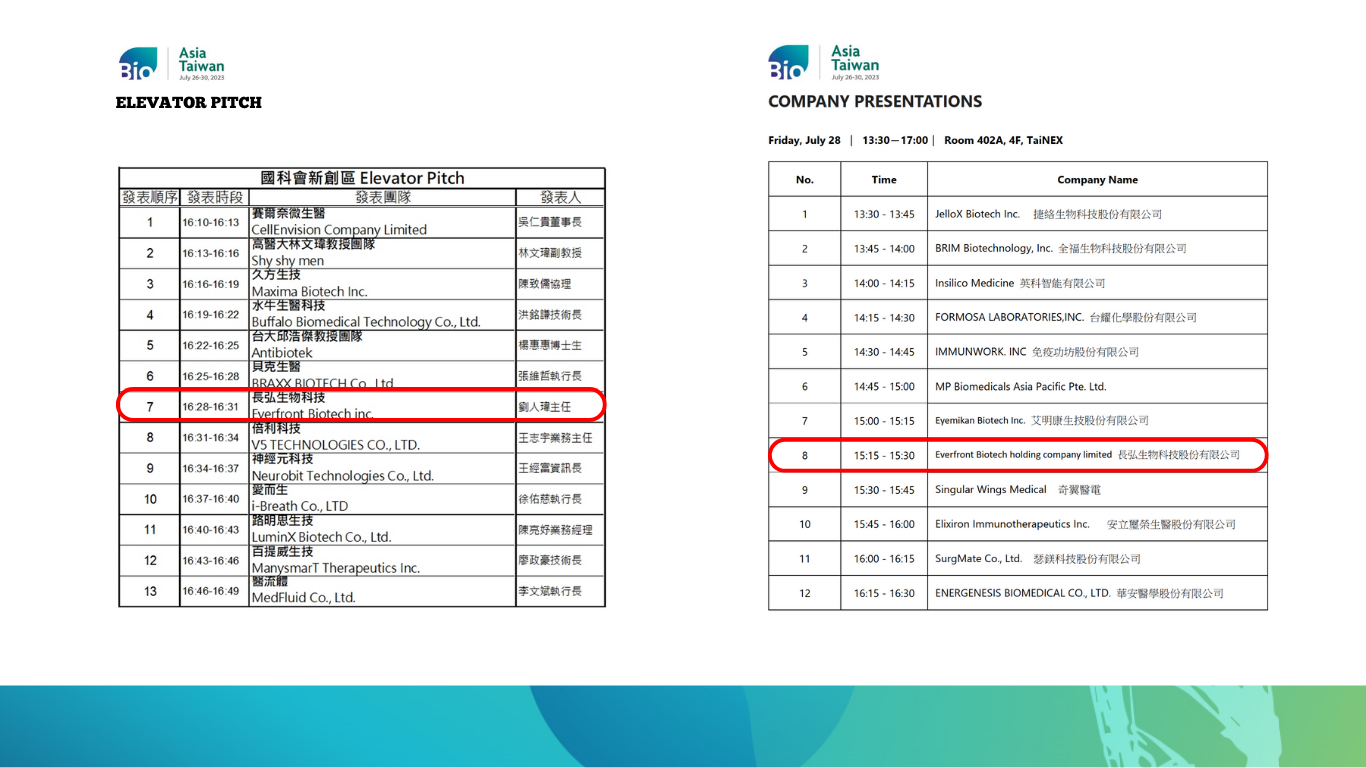
On the 27th, Everfront Biotech participated in the Precision Marketing (Elevator Pitch) organized by the National Science Council, followed by the Company Presentations on the 28th.

Live coverage of the “Elevator Pitch” on the afternoon of July 27th: Dr. Jen-Wei Liu, the Director of Clinical Medicine Research Department, presented Everfront Biotech’s most promising product, Cerebraca® Wafer, in a vivid and fast-paced manner.

On the morning of July 28th, during the corporate presentation, Dr. Jen-Wei Liu, the Director of Clinical Medicine Research Department, introduced the current status of new drug development at Everfront Biotech and provided an update on the latest clinical progress and results of Cerebraca® Wafer product. The session also included interactive engagement with the attending audience.

On the afternoon of July 28th, Dr. Jen-Wei Liu, the Director of Clinical Medicine Research Department, represented Everfront Biotech at the National Science Council’s Company Presentations. He provided a detailed explanation of the company’s current active process leading to Nasdaq listing, the main new drug development pipelines, and the results of clinical trials. His presentation also sparked engaging discussions and participation from numerous attendees.
On the afternoon of July 27th, the National Development Council’s New Business Incubation Program presented the “INNO ZONE Exhibition Selection Award.” This award aims to encourage biomedical teams and startups to actively engage with the international community in the early stages of development, increase the visibility of their technologies and products, and attract international funding and talents to accelerate growth. Therefore, Everfront Biotech was entrusted by the Biomedical Product Commercialization Center to participate in BIO Asia Taiwan 2023, enabling the seamless integration of international resources and a comprehensive presentation to potential investors.
Everfront Biotech is honored to have achieved the second place in the “INNO ZONE Exhibition Selection” and looks forward to showcasing the clinical trial results of Cerebraca® Wafer to the visitors, in the hope of generating keen interest and willingness for investment.

On the 27th, Everfront Biotech was awarded the second place in the “INNO ZONE Exhibition Selection” by the National Biotechnology Park. The award was presented by Director Li-Ju Wang of the Biomedical Product Commercialization Center (pictured second from the left), and received by Director Jen-Wei Liu of our Clinical Medicine Research Department (pictured second from the right) on behalf of our company.

Highlights of BIO ASIA-Taiwan 2023 Exhibition

Highlights of BIO ASIA-Taiwan 2023 Exhibition
Everfront Biotech Holding Company Limited - Brain Cancer Clinical Programs and Research Pipelines
Product Highlights of Cerebraca® Wafer:
Everfront Biotech’s innovative new drug, Cerebraca® Wafer, is a precision medicine sustained-release wafer that revolutionizes the treatment of malignant brain cancer. Through multiple patented novel features, Cerebraca® Wafer has successfully addressed the challenges of treating malignant brain cancer, which is difficult to manage and prone to relapse. Chemotherapy gradually loses its effectiveness against brain cancer cells due to their drug resistance, and the high proportion of cancer stem cells further exacerbates the problem of cancer recurrence and treatment resistance. However, these issues can be addressed by the pharmacological mechanism of action of Cerebraca® Wafer. In addition, malignant brain cancer cells express PD-L1, which renders the immune system unable to kill cancer cells. The successful development of Cerebraca® Wafer has also addressed this issue. In Phase I clinical trials, Cerebraca® Wafer has demonstrated efficacy without any drug-related adverse reactions. The median or average cumulative survival of relapsed malignant brain cancer patients has reached 25-26 months, which is significantly better than existing strategies such as Gliadel® wafer implantation (6.4 months), Temodal capsules (5.8 months), or Avastin injection (9.4 months). Cerebraca® Wafer has been approved by the Taiwan Ministry of Health and Welfare and the Human Research Ethics Committee of Hualien Tzu Chi Hospital for compassionate use trials. For malignant brain cancer patients who cannot undergo surgery and have tumors growing at the cervical spine, implanting Cerebraca® Wafer through a small incision has resulted in tumor shrinkage. In addition, combining Cerebraca® Wafer with cytokine-induced killer cells (CIK) for treatment can achieve a recurrence-free survival period of more than 40 months. With its outstanding clinical results, Everfront Biotech Inc. is actively seeking partners to jointly develop this new drug and help more malignant brain cancer patients.
Introduction to Everfront Biotech Co., Ltd:
Everfront Biotech Inc. was founded in 2010, with a focus on research and development of new drugs, specifically the screening and further development of potential small molecules for clinical study. The company’s team is responsible for all aspects of product development, from front-end potential product development to pre-clinical research, chemistry manufacturing and controls (CMC), clinical trial submissions, and clinical trial planning and execution. To date, the Everfront Biotech team has successfully applied three clinical trials: Cerebraca® Wafer (for malignant brain tumors), HK-001 (for amyotrophic lateral sclerosis), and EF-009 (for pancreatic cancer), which have been approved for IND applications in both the United States and Taiwan.
Everfront Biotech’s new drug development is in line with national policies, and the Cerebraca® Wafer and HK-001 products were granted the certifications of biotech and new drug in 2016. The company has received the government subsidies in three science and technology projects as of 2022 from the Ministry of Economic Affairs.