The Research Team for Developing a New Drug, EF-009, for the Treatment of Pancreatic Cancer Won the National Innovation Award
Date: November 16, 2021

Left to right: Dr. Tzyy-Wen Chiou, Director of R&D at National Dong Hwa University; Dr. Shinn-Zong Lin, Superintendent of Hualien Tzu Chi Hospital; Dr. Pei-Wen Chou, Chairwoman of Everfront Biotech Inc.; Dr. Horng-Jyh Harn, Vice R&D Director and Professor; and Dr. Jen-Wei Liu at Everfront Biotech Inc. The group won the National Innovation Award for the development of the drug EF-009 for treating pancreatic cancer.
The Tzu Chi Medical Foundation’s Innovation and Research Center, along with the Tzu Chi Hospital in Hualien, have developed a new drug called EF-009 for treating pancreatic cancer, using a targeted sustained-release delivery method. The drug has obtained patents in several countries, including the US, Japan, and the EU, and has received approval for clinical trials by the US Food and Drug Administration (FDA) and Taiwan’s Ministry of Health and Welfare. It has also been recognized with the “Academic Research and New Venture Award” at the National Innovation Awards this year.
Led by President Shinn-Zong Lin of the Innovation and Research Center, along with Professor Horng-Jyh Harn and Director Tzyy-Wen Chiou of Dong Hwa University’s Office of Research and Development, the research team’s work has been awarded the “Academic Research and New Venture Award” at the National Innovation Awards for six consecutive years.
In 2019, the team discovered that the small-molecule drug EF-API-001 could inhibit the cancer immune checkpoint PD-L1, activate immune T cells, and enhance the effectiveness of immunotherapy for malignant glioblastoma, which was a breakthrough in global immunotherapy research for malignant brain tumors. This research was also recognized with the “Academic Research and New Venture Award” at the National Innovation Awards. Another targeted drug called Cerebraca® wafer for the treatment of glioblastoma multiforme has also entered phase II trials.
Professor Horng-Jyh Harn explained that the treatment of glioblastoma multiforme and pancreatic cancer is a very difficult challenge in clinical. In the case of pancreatic cancer, the primary reason for its occurrence is the formation of a desmoplastic stroma, which creates a fibrosis protective layer that blocks chemotherapy drugs. Current pancreatic cancer drugs are unable to penetrate this layer, and the treatment results are poor, with a median survival time of only 6 to 8 months and a five-year survival rate of only 5%, which cannot meet the medical needs. Effective pancreatic cancer drugs are still lacking in clinical practice.
Compared to clinical chemotherapy drugs, Gemcitabine has been shown to prolong the lifespan of pancreatic cancer animal models by about 2 weeks in past experiments. Professor Horng-Jyh Harn said that in animal experiments targeting pancreatic cancer treatment, the new drug EF-009 can effectively prolong the lifespan of test animals by about 2.2 times. In the future, an orally administered drug with a synergistic effect will also be developed.
Professor Horn-Jyh Harn pointed out that the new pancreatic cancer drug EF-009 is implanted through endoscopic surgery and attached to the surface of the pancreas. It utilizes the characteristics of high concentration, high permeability, and long duration of local drug delivery, slowly releasing over a month. It penetrates the tumor matrix fibrosis protective barrier and inhibits the cancer cell immune checkpoint PD-L1, which turns off the “brake” that suppresses immune T cells against cancer cells. After activation, T cells can recognize tumors and kill tumor cells, thereby improving the effectiveness of immunotherapy. This is the first pancreatic cancer drug developed in Taiwan, with multiple international patents and technology transfer to Everfront Biotech Inc.
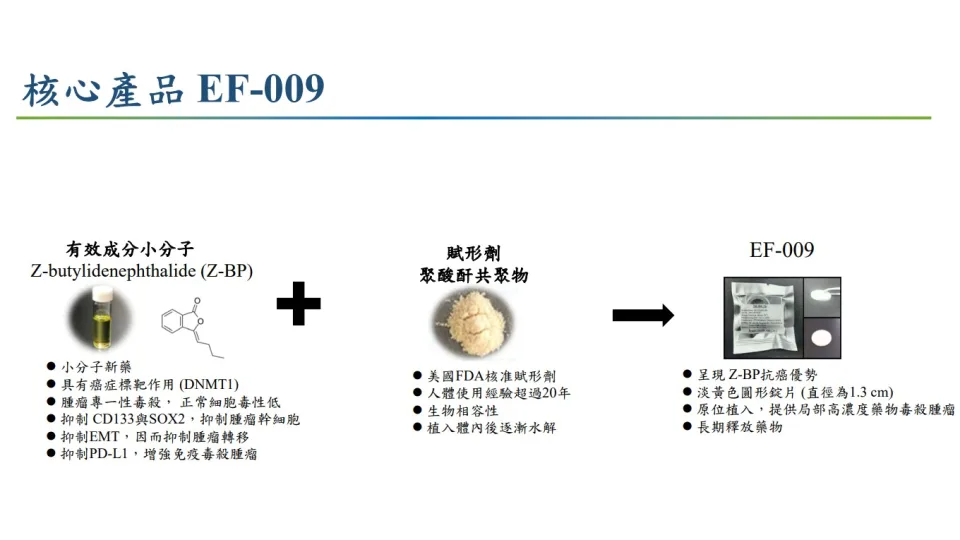
Professor Horng-Jyh Harn pointed out that after the implantation of EF-009 wafer targeted drug attaches on the pancreas for one month, followed by conventional treatment and in combination with oral TS-1, PD-L1 can be continuously inhibited, allowing T cells to recognize and kill pancreatic cancer cells. The target of this targeted therapy is to target AMP activated protein kinase (AMPK) in DNA methyltransferase (DNMT), which is involved in epigenetic regulation.
The preclinical development of EF-009 has been completed, and the process meets the international standards of PIC/S GMP for the production of active pharmaceutical ingredient, excipient, and drug product – Cerebraca® wafer. Results from toxicity studies, safety pharmacology studies, genotoxicity studies, and pharmacokinetic studies have preliminary confirmed its safety and drug metabolism characteristics.
This new drug has been transferred to Everfront Biotech Inc. and has been granted Investigational New Drug (IND) approval by the US FDA and Taiwan’s Ministry of Health and Welfare. It is expected to enter Phase I/IIa trial to test its safety and efficacy in humans, with the hope of helping pancreatic cancer patients who require treatment in the future.
Professor Horng-Jyh Harn also stated that in addition to continuing new drug development, the research results related to the new drug EF-009 will also be presented and exhibited at the Taiwan Healthcare+ Expo from December 2 to 5.