Pancreatic Cancer

Pancreatic Cancer (PC)
Pancreatic cancer often presents with subtle early symptoms, leading to frequent misdiagnosis. As the disease progresses to advanced stages, symptoms become more severe and pronounced, making treatment significantly challenging with limited effectiveness.
Over the past 20 years, the diagnosis rate of pancreatic cancer in Taiwan has increased more than threefold, and the mortality rate has increased by 1.7 times. Each year, more than 2,500 new cases are reported, and pancreatic cancer ranked 7th among the top ten deadliest cancers in Taiwan in 2021, with a mortality rate of 11.3%. The five-year survival rate for unresectable pancreatic cancer is less than 5%.
According to the International Agency for Research on Cancer (IARC), in 2018, there were 451,000 global diagnoses of pancreatic cancer, resulting in 440,000 deaths. The 5-year survival rate for pancreatic cancer worldwide is less than 10%.
According to the latest Cancer Facts & Figures 2025 report by the American Cancer Society (ACS), in 2025, the United States is expected to have 67,440 new cases of pancreatic cancer, and an estimated 51,980 people will die from the disease. Pancreatic cancer is the 10th most common cancer in the United States, but it ranks 3rd in terms of mortality rate, only behind lung cancer and colorectal cancer. The five-year survival rate for pancreatic cancer in the United States is approximately 13%, which remains the lowest among all cancers.
Causes of Pancreatic Cancer
Changes in DNA can potentially lead to cancer, and these changes may be inherited from your parents or may develop over time. The latter changes might be caused by exposure to harmful substances, but they can also occur purely by chance. To date, the exact causes of pancreatic cancer remain unclear. Approximately 10% of cases are considered familial or hereditary, while most pancreatic cancers occur randomly or are induced by factors such as smoking, obesity, and age.
If a direct relative of yours is diagnosed with pancreatic cancer, your own risk of developing the disease may also increase. Therefore, it is recommended that family members of pancreatic cancer patients undergo genetic mutation testing. Dr. Lin reminds us that having one family member with pancreatic cancer increases the relative risk by 4.5 times. If there are two family members, the risk increases to 6.4 times, and with three family members, the risk can be as high as 32 times. Additionally, the age of onset tends to be relatively younger, potentially under 50 years old (refer to the chinese news).
In addition, there are other factors that may increase an individual’s risk of developing pancreatic cancer:
- Long-term diabetes
- Chronic and hereditary pancreatitis
- Genetic mutations (such as BRCA1 or BRCA2 genes)
- Smoking
- Ethnicity
- Age (over 60)
- Gender (men have a slightly higher risk)
- Regular consumption of red and processed meats
- Obesity
- The Risk (Potential Risk) of Long-Term Consumption of Sugary Beverages: Recently, a report highlighted the case of a woman in her 50s who discovered that her CA19-9 tumor marker level was over 40 times higher than the normal range during a blood test. After consulting with her doctor and making dietary adjustments, it was confirmed that her daily consumption of sweetened black tea was the cause of the abnormal marker. The woman had been drinking 1000 to 2000 mL of black tea every day for years, and after stopping, her CA19-9 levels quickly returned to normal. Ultrasound and other tests confirmed no presence of cancer cells. However, her doctor advised against the long-term consumption of sugary beverages to prevent the potential risk of pancreatitis or increased cancer risk.
- Pancreatic cysts: Research indicates that approximately 10% of pancreatic cancer cases are associated with pancreatic cysts. The cysts most closely linked to malignancy are mucinous cystic neoplasms (MCN) and intraductal papillary mucinous neoplasms (IPMN). Regular MRI monitoring and early diagnosis by a physician are crucial. If the cysts present a high risk, surgical removal is often recommended to reduce the potential for future cancer development.
It is important to note that the presence of these risk factors does not guarantee that an individual will develop pancreatic cancer, nor does every person with pancreatic cancer necessarily have these risk factors. This information is intended for general health maintenance and reference.
Background of Pancreatic Cancer

- Lack of effective treatment options: Treatment methods for pancreatic cancer include surgical resection, radiation therapy, and chemotherapy, but they all have certain limitations and challenges. Surgical resection is the preferred method for treating pancreatic cancer, but it can only be performed when the cancer is diagnosed early. Radiation therapy and chemotherapy can be used for advanced or metastatic pancreatic cancer, but their effectiveness is limited and can lead to side effects.
- Difficult to Detect Early, High Mortality Rate: Due to the pancreas’ location behind the spine in the retroperitoneal space, pancreatic cancer often presents no symptoms in its early stages. Consequently, many patients are diagnosed at an advanced or terminal stage, making complete surgical removal challenging. Additionally, the malignant nature of pancreatic cancer cells, which are prone to metastasis and recurrence, contributes to its high mortality rate. Research indicates that the five-year survival rate for stage I pancreatic cancer can reach 90%, but it drops to just 13% by stage IV, highlighting the importance of early detection and treatment.
- Notable individuals affected: Many well-known figures both domestically and internationally have succumbed to pancreatic cancer, including Steve Jobs, the co-founder of Apple Inc., French chef Paul Bocuse, and one of the “Three Tenors,” Luciano Pavarotti.
- Diagnosis: Due to its location in the retroperitoneal cavity and its elongated glandular structure with both endocrine and exocrine functions, the pancreas is difficult to evaluate fully using ultrasound. Combined with the fact that pancreatic cancer often lacks obvious symptoms in its early stages and is surrounded by numerous blood vessels, once the cancer cells invade the surrounding vessels, it becomes inoperable.
High Risk Groups and Early Symptoms of Pancreatic Cancer

- High-Risk Groups: Risk factors for pancreatic cancer include high-sugar and high-fat diets, smoking, alcohol consumption, genetics, diabetes, obesity, and chronic pancreatitis. According to National Health Insurance data, type 2 diabetes ranks as the leading comorbidity associated with pancreatic cancer, indicating that diabetic patients should be particularly vigilant. Additionally, those who suddenly develop diabetes should be cautious, as this may be a sign of pancreatic cancer affecting insulin secretion and leading to elevated blood sugar levels.
- Dr. William Fisher has developed an algorithm to track how many newly diagnosed diabetes patients over the age of 50 will develop pancreatic cancer within three years. The diagnosis of diabetes not only correlates with an increased risk of pancreatic cancer, but the time interval between diabetes diagnosis and cancer development may also allow for early intervention. Additionally, this research aims to identify suitable biomarkers for better detection and treatment (original source).
- A large-scale study across four continents involving nearly 2.5 million participants found that every 10 g/day increase in alcohol intake was linked to a 3% higher risk of pancreatic cancer. Risk was more pronounced in women consuming ≥15 g/day and men ≥30 g/day. Beer and spirits showed stronger associations than wine. No clear link was found in Asian cohorts, suggesting drinking patterns may influence risk. The findings support a modest but significant positive association between alcohol and pancreatic cancer.
- Early symptoms to watch for: upper abdominal bloating, upper abdominal pain, weight loss (due to poor appetite, indigestion), changes in stool (oily, pale color, foul smell, difficulty flushing), diarrhea (persistent for several days), Jaundice (tea-colored urine, itchy skin, yellowing of the whites of the eyes), back pain, elevated blood sugar (more common in late-stage patients), and a recent diagnosis of diabetes last year.
- Additionally, before a diagnosis of pancreatic cancer, there may be an increase in body temperature (known as neoplastic fever), characterized by recurrent and persistent fever without symptoms of infection. There may also be a decrease in cholesterol and triglyceride levels due to reduced fat absorption by the body, which can lead to gradual weight loss.
- Blood Clots: A blood clot may form within a vein without any accompanying bleeding. In pancreatic cancer patients, clots can also develop in veins near the pancreas, such as the splenic vein or portal vein. This condition is known as deep vein thrombosis (DVT) and can disrupt normal blood flow in the veins, leading to impaired circulation.
- Moreover, recent studies have indicated that under the influence of a high-fat diet and stress, stress-related neurotransmitters and obesity-related hormones activate a protein called cAMP response element-binding protein (CREB) through different biological pathways. This protein is associated with cancer cell growth, potentially accelerating the progression of pancreatic cancer. This finding is particularly noteworthy for pancreatic cancer patients (Journal).
Diagnosis of Pancreatic Cancer
To detect pancreatic cancer early, imaging examinations are still the more reliable method. Although serum tests (such as CEA and CA19-9 tumor markers) are not significant in the early stages and can only serve as a reference, they can still be chosen as one of the regular screening items.
Ultrasound
Ultrasound is a convenient diagnostic tool, but the presence of gas in the adjacent gastrointestinal tract can interfere and make it challenging to achieve comprehensive assessments of the pancreas.
Computed Tomography (CT) or Magnetic Resonance Imaging (MRI)
Both imaging techniques have the drawback of sometimes being unable to identify tumors smaller than 2 centimeters, which accounts for about 1/3 to 1/4 of cases. Additionally, some patients may be allergic to contrast agents used in these procedures.
A research team led by Professor Wei-Chih Liao at National Taiwan University Hospital has developed an AI-assisted diagnostic system named “PPANCREASaver.” This system enhances the detection accuracy of pancreatic tumors smaller than 2 centimeters using CT imaging. In 2022, it received the Breakthrough Device designation from the U.S. FDA and has since been certified by the Taiwan Food and Drug Administration (TFDA). Currently, PPANCREASaver is available as a self-paid service at National Taiwan University Hospital.

Endoscopic Ultrasound
This method has the highest sensitivity for detecting tumors smaller than 1 centimeter, but it is an invasive procedure. The ultrasound probe must be passed through the intestinal wall to reach the pancreas, which carries some risks, making it a non-first-line diagnostic approach. However, compared to CT/MRI, which have high sensitivity and can simultaneously perform biopsy examinations, they are also more convenient.

Blood Tests – CEA and CA19-9 Tumor Markers
These blood tests can be used as risk indicators. However, elevated levels of these markers typically occur only in advanced-stage pancreatic cancer and can be affected by factors such as liver, kidney, and cystic diseases, leading to potential misinterpretations. Therefore, they can only serve as reference points.
Breath Test (Clinical Trial Underway)
Targeting the challenge of difficult early detection in pancreatic cancer, a groundbreaking “breath test” clinical trial, led by Professor George Hanna and funded by Pancreatic Cancer UK, is currently ongoing (launched in 2025). The test works by analyzing a specific combination of Volatile Organic Compounds (VOCs) in the breath, serving as unique biomarkers for identifying pancreatic cancer patients. The goal of this technology is to serve as a preliminary screening tool, allowing high-risk individuals to be quickly referred for confirmation using existing standard diagnostic tests. This development is expected to fundamentally and significantly improve the rate of early diagnosis for pancreatic cancer.
Current Treatment Options for Pancreatic Cancer

Chemotherapy
Chemotherapy uses anticancer drugs to kill cancer cells, but the dense stromal tissue surrounding pancreatic cancer makes it difficult for the drugs to penetrate effectively, resulting in relatively lower efficacy. Chemotherapy often employs combination therapy to enhance effectiveness. For instance, a clinical study involving previously untreated metastatic pancreatic adenocarcinoma patients showed that the overall survival period for those receiving combination therapy with nab-paclitaxel + gemcitabine was 8.5 months, compared to 6.7 months for the gemcitabine-alone group. This combination therapy is considered less toxic than FOLFIRINOX and serves as a frontline chemotherapy option. However, chemotherapy may cause side effects such as nausea, vomiting, hair loss, and fatigue. It remains one of the main treatment modalities for pancreatic cancer.
Surgical Resection
When the size of the pancreatic tumor is smaller than 2 centimeters, more than 80% of patients are candidates for surgical treatment, significantly improving their prognosis. The practicality of surgery depends on tumor size, location, proximity to nearby blood vessels, and whether there is distant metastasis. However, only about 20% of newly diagnosed patients are deemed suitable for resection.
Current Treatment Reference Drugs
Due to the limited efficacy of current drugs and their limited impact on patient survival and quality of life, many researchers are seeking more effective medications and treatment methods.
Pancreatic cancer can be treated without chemotherapy. Some patients might opt out of chemotherapy due to its severe side effects or because their health condition is not suitable. Alternative treatments include surgery, radiation therapy, targeted therapy, and immunotherapy. Surgery can remove the tumor, radiation can shrink tumors or alleviate symptoms, and targeted and immunotherapy often have fewer side effects. Additionally, supportive therapies like pain management and nutritional support can improve quality of life. Patients should discuss with their doctors to decide the best treatment plan.
Onivyde® (Irinotecan Liposomal Injection)
Onivyde® is a nanoliposomal injection containing Irinotecan, a camptothecin derivative. Its mechanism of action is as a topoisomerase I inhibitor, a cytotoxic drug that has been approved and widely used for cancer treatment.
It is indicated for metastatic pancreatic cancer in combination with 5-fluorouracil and leucovorin for patients who have previously received gemcitabine.
Clinical trial data from 2016 revealed that using Onivyde® in combination therapy for advanced pancreatic cancer increased the one-year survival rate to 26%, a 63% improvement compared to the use of 5-fluorouracil/leucovorin alone.
- Onivyde was included in Taiwan’s National Health Insurance coverage in 2018 and is also marketed in the United States, Europe, China, and Japan.
Potential side effects may include severe neutropenia, severe diarrhea, interstitial lung disease, and allergic reactions.
Gemzar® Injection (Gemcitabine HCl)
- Gemzar® is an intravenous injection containing gemcitabine and is used in the treatment of non-small cell lung cancer, pancreatic cancer, bladder cancer, and other cancers. It is employed as a first-line treatment for inoperable stage II or III pancreatic cancer and metastatic stage IV pancreatic cancer. It can also be used in patients who have received prior treatment with 5-FU.
- Its mechanism of action involves blocking DNA replication and cell division in cancer cells, thereby inhibiting their growth and spread. According to clinical trial reports, the overall median survival period with Gemzar® was 5.7 months, compared to 4.2 months for the control group treated with fluorouracil.
- Potential side effects include nausea, vomiting, rash, and fever.
TS-1® Capsule (Tegafur、Gimeracil、Oteracil potassium Capsules)
- S-1 is an oral capsule containing Tegafur, Gimeracil, and Oteracil potassium, with the main anticancer component being 5-FU. The primary mode of action of 5-FU is through competitive binding of its active metabolite FdUMP with dUMP and thymidylate synthase, simultaneously forming a trimer with reduced folate to inhibit DNA synthesis.
- S-1 is mainly used to treat gastric cancer, pancreatic cancer, colorectal cancer, advanced non-small cell lung cancer, and biliary tract cancer. Common side effects include constipation, diarrhea, poor appetite, nausea, and vomiting. Clinical trial data showed that the median overall survival period for the S-1 monotherapy group was 9.66 months, similar to the Gemcitabine monotherapy group (8.80 months).
- Patients receiving S-1 + Gemcitabine combination therapy had a median overall survival period of 10.05 months, longer than the Gemcitabine monotherapy group (8.80 months).
BIZENGRI® (zenocutuzumab-zbco)
- In December 2024, the FDA granted accelerated approval for BIZENGRI® for the treatment of pancreatic cancer patients with Neuregulin 1 (NRG1) gene fusion mutations. Approximately 3% of pancreatic cancer patients exhibit this alteration, also known as NRG1 fusion positive.
- Clinical trials conducted by Merus showed that 40% of the 30 pancreatic cancer patients with NRG1 gene fusion experienced tumor reduction. Among those who responded, two-thirds had their response last for at least six months.
- The most common side effects include diarrhea, musculoskeletal pain, fatigue, nausea, infusion-related reactions, difficulty breathing, rash, constipation, vomiting, abdominal pain, and edema.
>> Source: News Article
Current Medical Device Therapies

Optune Pax®
Tumor Treating Fields (TTFields) is a portable, physical cancer therapy that utilizes alternating electric fields tuned to specific frequencies to selectively target and kill tumor cells through multiple mechanisms. Its key mechanisms include:
- Interfering with mitosis
Downregulating DNA damage repair
Enhancing downstream anti-tumor immunity
Interfering with tumor cell migration
- The U.S. FDA has officially approved Novocure’s Optune Pax® (Tumor Treating Fields technology) in combination with standard chemotherapy as a first-line treatment for locally advanced pancreatic cancer.
According to data from the Phase 3 PANOVA-3 clinical trial, it improved the median overall survival (mOS) by 2 months compared to patients receiving gemcitabine/nab-paclitaxel alone. Furthermore, it significantly extended the patients’ “Time to pain progression” from 9.1 months to 15.2 months, without increasing systemic toxicity.
The most common side effect is localized skin reactions, with approximately 76% of patients experiencing erythema, itching, or inflammation at the array placement sites.
(This image is for illustrative purposes only and does not represent the actual product appearance.)
Investigational Drugs
SM-88 <Phase II/III>
SM-88 (tyrosine derivative [Td], mTOR inhibitor, CYP3a4 inducer, and oxidative stress catalyst) developed by Tyme Technologies is a targeted therapy with relatively low toxicity, leveraging the Warburg Effect along with oxidative stress to induce tumor cell death.
Its excellent suitability for pancreas cancer lies in its capacity to penetrate tumors and be well-tolerated by debilitated patients.
This drug development was terminated in 2021 due to the failure to meet the primary endpoint of overall survival in the Phase II/III clinical trial (NCT03512756).
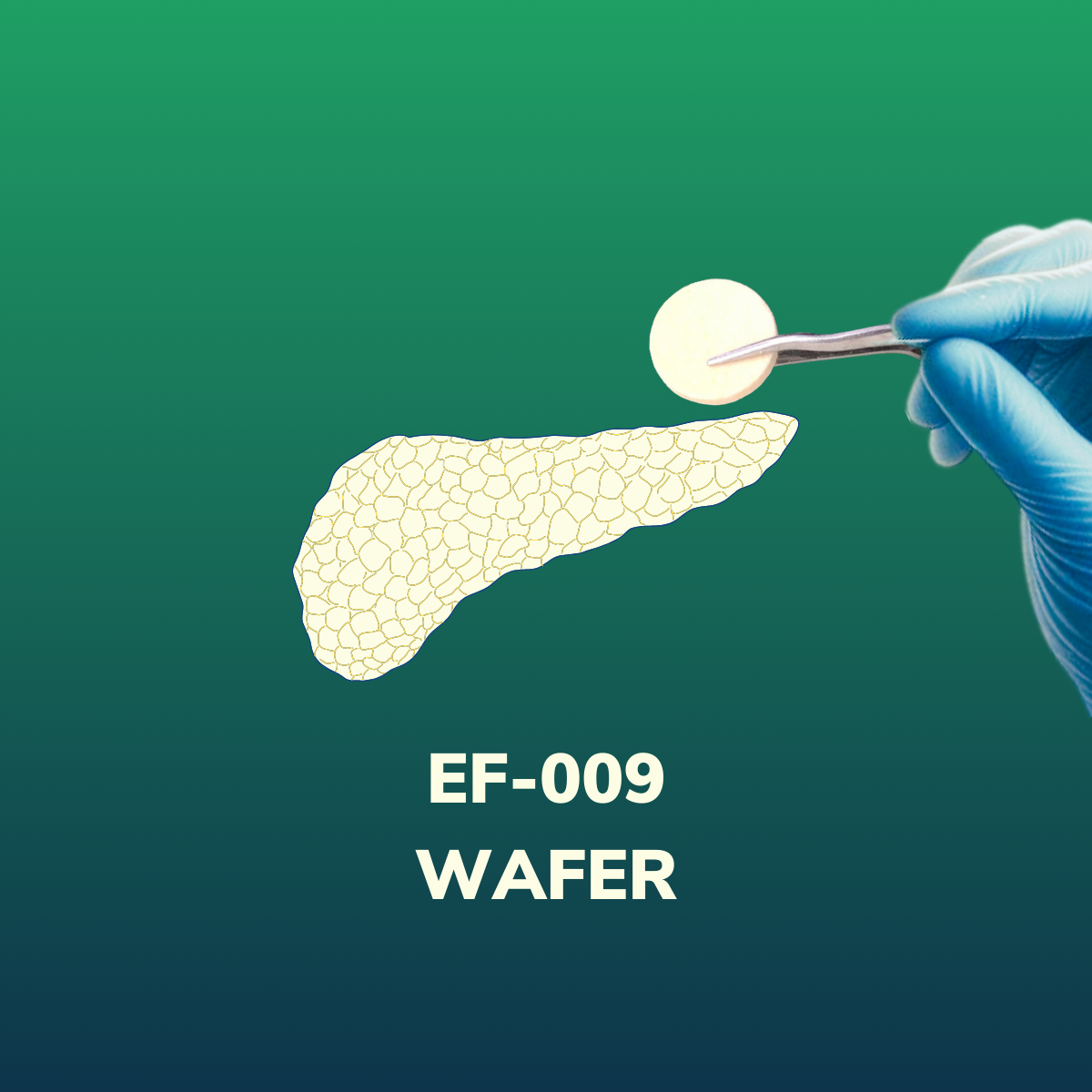
EF-009 Wafer <Phase I> >>more
The mortality rate of pancreatic cancer (PC) is rising rapidly.
However, the treatment of pancreatic cancer remain a formidable challenge and has 5-year survival rate of approximately less than 5%. Surgery followed by adjuvant chemotherapy the standard of care – without additional therapy, over 90% of patients relapse and die. However, PC remains highly resistant to conventional chemotherapy.
The great challenges lies firstly, in delivering chemotherapeutic agents effectively to bypass the protective barriers around pancreatic cancer; and secondly, in reversing the immunosuppressive environment caused by the presence of cancer stem cells.
EF-009 Wafer developed by Everfront Biotech Inc. is a slow-releasing drug for the treatment of pancreatic cancer with multiple targeting effects. It can accumulate in targeted areas around the tumor, make it vulnerable and easily killed by chemotherapy or immune cells, with manageable toxicity.
More Investigational Drug Information
Revolution Medicines: On September 10, 2025, the company announced the latest Phase 1 clinical results of its RAS(ON) multi-selective inhibitor daraxonrasib (RMC-6236). The results showed that both monotherapy and combination with chemotherapy demonstrated therapeutic efficacy in patients with metastatic pancreatic ductal adenocarcinoma (PDAC). In second-line and later patients, daraxonrasib monotherapy achieved an objective response rate (ORR) of 29–35%, a disease control rate (DCR) of 92–95%, a median progression-free survival (mPFS) of about 8 months, and a median overall survival (mOS) of 13–15 months. In first-line patients, daraxonrasib monotherapy achieved an ORR of 47%, a DCR of 89%, with good tolerability. Meanwhile, the combination regimen with gemcitabine and nab-paclitaxel (GnP) showed even greater efficacy, with an ORR of 55% and a DCR of 90% (source: news).
- Immuneering: In June 2025, the company announced results from a Phase 2a clinical trial evaluating first-line treatment for metastatic pancreatic ductal adenocarcinoma (mPDAC). The study showed that the MEK inhibitor atebimetinib, in combination with gemcitabine and nab-paclitaxel, demonstrated promising therapeutic potential. At a daily dose of 320 mg, the 6-month overall survival rate reached 94%, and the progression-free survival rate was 72%, both exceeding historical benchmarks. The objective response rate (ORR) was 39%, and the disease control rate (DCR) reached 81%. Additionally, the combination therapy exhibited a favorable safety profile (source: news).
- Actuate Therapeutics: In June 2025, Actuate announced the results of a Phase II clinical trial involving 286 patients with metastatic pancreatic ductal adenocarcinoma (mPDAC), evaluating the efficacy of the GSK-3β inhibitor elraglusib. The study showed that the median overall survival in the elraglusib treatment group was 10.1 months, significantly longer than 7.2 months in the control group, successfully meeting the trial’s primary endpoint (source: news).
- Novocure: In August 2025, Novocure submitted a Premarket Approval (PMA) application to the FDA for Tumor Treating Fields (TTFields) in locally advanced pancreatic cancer, with a decision expected in the second half of 2026. The submission is based on results from the phase 3 PANOVA-3 trial (Source: News), which demonstrated that TTFields combined with standard chemotherapy significantly improved overall survival (16.2 months vs 14.2 months) and also prolonged pain-free survival and distant progression-free survival. Although no significant differences were observed in overall PFS, local PFS, or ORR, TTFields was well tolerated, with only mild to moderate skin-related adverse effects, and maintained patients’ quality of life (Source: News).
- BIOGATE PRECISION MEDICINE: BGX targeted therapeutic biologic works by blocking intracellular signal transduction to inhibit cell proliferation and spread. In animal experimental studies, BGX has demonstrated significant effects in inhibiting tumor growth and metastasis, while extending survival rates. The company will soon apply for TFDA clinical trials (Chinese News)
- EDAP TMS: Initiates the first I/II phase multicenter study named PULS. This clinical trial targets patients with locally advanced and unresectable pancreatic tumors, using High-Intensity Focused Ultrasound (HIFU) to ablate the pancreatic lesions (Source: News).
- High-Concentration Vitamin C Injection: According to a phase II clinical trial, high-dose Vitamin C IV (75g) as an adjunct therapy to the standard treatment for pancreatic cancer (gemcitabine and nab-paclitaxel) can extend the average survival time of patients with advanced pancreatic cancer from 8 months to 16 months, and progression-free survival (PFS) from 4 months to 6 months (Source: news)
More Investigational Inspection
- Alibaba: Alibaba’s Damo Academy has developed an AI pancreatic cancer screening system, “Damo Panda,” which recently received Breakthrough Device designation from the U.S. FDA, potentially accelerating its path to market. Using deep learning to analyze abdominal imaging, the system demonstrated 34.1% higher accuracy than radiologists in early testing. It is designed to detect pancreatic cancer in asymptomatic patients, aiming to improve early diagnosis and survival rates (Source: news)
Care and Considerations for Pancreatic Cancer Patients

Cachexia in Pancreatic Cancer
Pancreatic cancer has the highest prevalence of cachexia among all cancer types, affecting approximately 80% of patients. This condition leads to muscle wasting, malnutrition, reduced quality of life, and significantly shortened survival. Although progress has been made in cachexia research in recent years, its high prevalence remains a major clinical challenge. After diagnosis, early nutritional risk assessment and active intervention are essential, including stabilizing body weight, reducing disease burden, and improving quality of life. Future strategies should combine chemotherapy with nutritional support and explore non-pharmacological approaches such as exercise and acupuncture. A multimodal management strategy may help improve quality of life and extend survival in patients with pancreatic cancer (reference).

Nutritional Support
Pancreatic cancer patients often experience malnutrition and weight loss. This may be due to blockage of the main pancreatic duct by cancer cells, leading to inadequate secretion of pancreatic fluid into the duodenum. Undigested fats are then excreted in the feces, resulting in steatorrhea, chronic diarrhea, and weight loss. Adequate nutritional support is essential, and it is recommended to supplement with a high intake of proteins while controlling fruit consumption.
read more👉Balanced Diet for Cancer Patients

Very Low-Carb Diet Extends Advanced Pancreatic Cancer Survival by 34%
A breakthrough Phase II clinical trial published in the journal Cancer in March 2026 revealed that Stage IV pancreatic cancer patients combined standard chemotherapy with a “medical-grade very low-carbohydrate diet (< 30g daily)” to effectively cut off the tumor’s glucose supply. The results showed that the overall survival (OS) of the nutritional intervention group reached 13.7 months, a substantial 34% increase compared to the control group. Furthermore, progression-free survival (PFS) significantly improved to 8.5 months without increasing chemotherapy side effects, successfully preserving the patients’ quality of life.
Pancreatic Enzyme Supplementation
Patients with pancreatic cancer may be unable to secrete the enzymes necessary for digestion due to partial removal of the pancreas or pancreatic dysfunction. When the pancreas cannot produce enough enzymes to break down food, pancreatic enzyme supplements are needed. Discussing supplementation with a doctor or nutritionist can help improve the digestion and absorption of food.
If you experience poor digestion, a feeling of excessive gas in the abdomen, foul-smelling stools, floating stools with fat, abnormally colored stools, diarrhea, and weight loss, you should discuss taking digestive enzyme supplements with your doctor.
Pain Management
The pain and discomfort caused by pancreatic cancer are often due to chronic upper abdominal or back pain, which can worsen in the advanced stages of cancer, leading to “total pain.” Effective pain relief medication and other therapeutic measures are necessary to maintain a good quality of life. Patients should follow their physician’s instructions to alleviate the burden caused by pain. During hospitalization, the pain level for patients can reach as high as 10 (equivalent to labor pain), and the relief provided by oral or injectable pain medications may be limited. Consequently, patients can consider nerve block procedures as recommended by their doctor to significantly improve the impact of pain on their quality of life.

Psychological Support
The course of pancreatic cancer treatment may bring ongoing stress and anxiety to patients. They require continuous psychological support and care from family and friends. If needed, patients can seek the assistance of psychologists, mental health professionals, or even spiritual care providers to maintain a positive psychological state.
In the terminal stage of pancreatic cancer, patients may die due to liver failure, severe infections, bowel obstruction, malnutrition and weakness, intense pain, and multiple organ failure. These conditions lead to the accumulation of toxins in the body, immune system failure, severe abdominal pain and vomiting, and decreased strength and immunity, ultimately resulting in death. During this stage, it is crucial to manage pain and provide palliative care to reduce suffering and improve the quality of life.

Regular Follow-Up Examinations
After pancreatic cancer treatment, patients need regular check-ups to monitor tumor recurrence and other health issues. These examinations may include abdominal ultrasounds, computed tomography (CT) scans, or magnetic resonance imaging (MRI). Regular follow-up is crucial for effective disease management.

Postoperative Care
After undergoing surgical treatment, patients should adhere to their physician’s recommendations and undergo regular check-ups and rehabilitation. This may include physical therapy, recovery training, and other therapeutic approaches. Additionally, postoperative dietary adjustments usually start with a liquid diet, gradually progress to a soft diet, and finally, include regular solid foods.
Reference
- http://www.tsim.org.tw/journal/jour33-2/03.PDF
- https://www.pharmaengine.com/api/rd_downloadfile.ashx?guid=E134C481-1BC5-4590-8947-C6E9C00B427E
- https://info.fda.gov.tw/mlms/ShowFile.aspx?LicId=02023298&Seq=087&Type=9
- https://ch.tty.com.tw/rwd1181/store/F3/TS-1.pdf
- https://www.cancer.org/cancer/types/pancreatic-cancer/about.html