HK-001 | ALS Treatment


Curing Rare Diseases
- Rare Diseases affect so few people that trying to cure them makes little economic sense without public incentives and special regulatory benefits.
- Our mission is to develop orphan drugs to treat rare diseases chosen for their low probability of survival due to the lack of effective treatments, the presence of few competitors offering objectively inferior products based on key medical metrics, and the possibility of rapidly gaining market share via licensing agreements with global pharma partners.
Amyotrophic lateral sclerosis (ALS)
The age of onset for amyotrophic lateral sclerosis (ALS) is 40 to 70 years, with a mean age of 55 years. In the United States, about 5,000 to 6,000 new cases are diagnosed each year, while in Taiwan, there are 300 to 500 new cases per year.
Approximately 90% of ALS cases have no clear family history or genetic mutation as the cause. The remaining 5-10% of cases are called familial ALS (FALS), which are inherited from known mutated genes associated with ALS.
No effective treatment: Currently: there is no known cure for ALS, and the only available treatment options can only slow the progression of the disease and improve quality of life.
High mortality rate: According to the statistics from the ALS Association, patients typically survive for an average of 2 to 5 years after onset of symptoms, which may vary from person to person, and only 10% of patients can survive more than 10 years.
Our Innovative Approach
Use Small Molecules Drugs as Our Key Therapeutics
Given their relatively low molecular weight, simple chemical structures, more predictable pharmacokinetics and pharmacodynamics, small molecule drugs not only have superior safety profiles, but they can pass through cell membranes to reach intracellular targets and be formulated in many forms.
Harness the Power of the Human Immune System
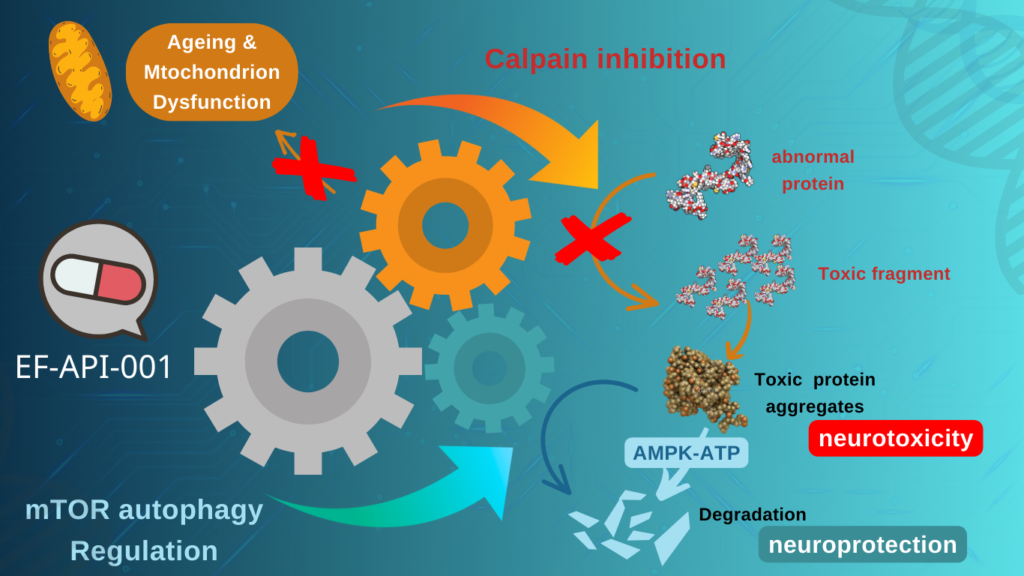
We are actively identifying and developing active compounds that can enhance the body’s natural defense capabilities across multiple indicators. Our goal is to achieve therapeutic effects by inhibiting the overactive autophagy process through mTor target inhibition and other mechanisms.
Improving Efficacy with Targeted Drug Delivery
We use targeted drug delivery platforms to improve the efficacy of our products with options like slow-released local delivery system, systemic drug delivery system, dripping pill formulation, dermal application formulation and injectable formulation.
Product Profile
Product Name
HK-001 Soft Capsules
Disease
Amyotrophic lateral sclerosis (ALS)
Target Market
The seven major countries, including the United States, France, Germany, Italy, Spain, the United Kingdom, and Japan, are expected to increase to 37,000 patients by 2023.
Market Size
In the 7 major countries is estimated to reach approximately USD 1.2 billion annually by 2027.
Competition Size
There is currently no drug on the market that can truly cure ALS.
Objective Size
First-in-class in the market, aiming to secure a 15% market share through direct competition.
HK-001 Soft Capsules
The HK-001 soft capsule contains a small molecule active pharmaceutical ingredient EF-API-001 produced under PIC/S GMP standards. Through targeted inhibition of mTor and suppression of excessive autophagy, it aims to delay the progression of ALS in patients.
- The HK-001 capsule is an oral dosage form, and two improvements were observed in animal testing results (ALS mice, SOD1G93A mice):
Delayed onset of ALS progression: Using a BBB scale (Basso, Beattie and Bresnahan scale) score of less than 15 as the disease onset threshold, it was observed that the onset time of ALS in mice orally administered with HK-001 was delayed compared to the control drug Riluzole.
Prolonged survival in ALS: The survival of ALS mice orally administered with HK-001 was superior to the untreated group and also superior to the control drug Riluzole.
To Determine the Maximum Tolerated Dose (MTD) of HK-001 in Healthy Volunteers
Phase I clinical Trial
- Initiated Phase I clinical trial in April 2021.
The Advantages of HK-001 in the Treatment of ALS are as follows:
Delayed Onset of ALS Progression
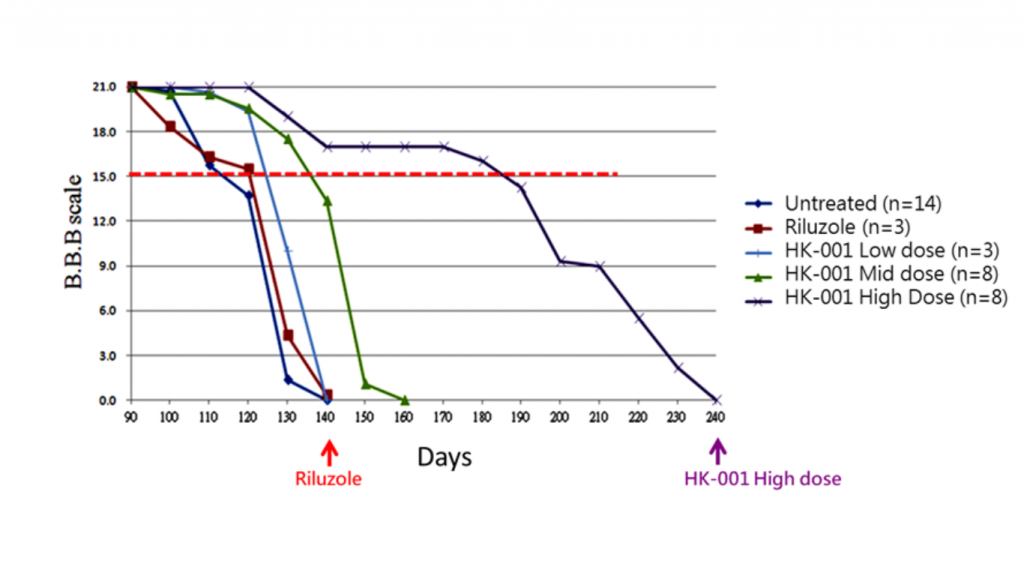
The number of Days with BBB scale < 15
Using a BBB scale score of less than 15 as the disease onset threshold, it was observed that the onset time of ALS in mice orally administered with HK-001 was delayed compared to the control drug Riluzole.
Prolonged Survival in ALS
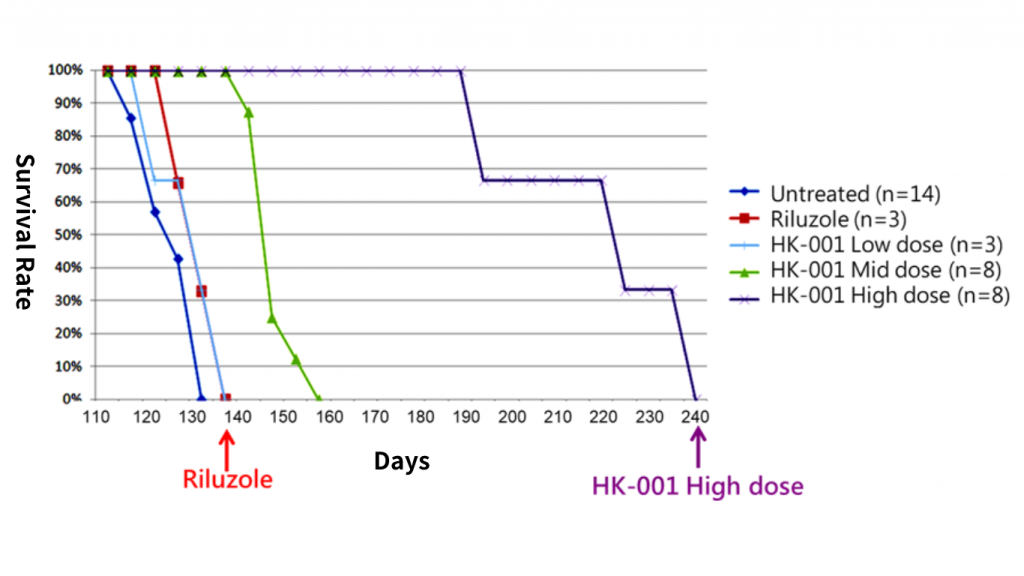
Survival
The survival of ALS mice orally administered with HK-001 was superior to the untreated group and also superior to the control drug Riluzole.
The Mechanism of Action of HK-001: Involves the Inhibition of Excessive Autophagy
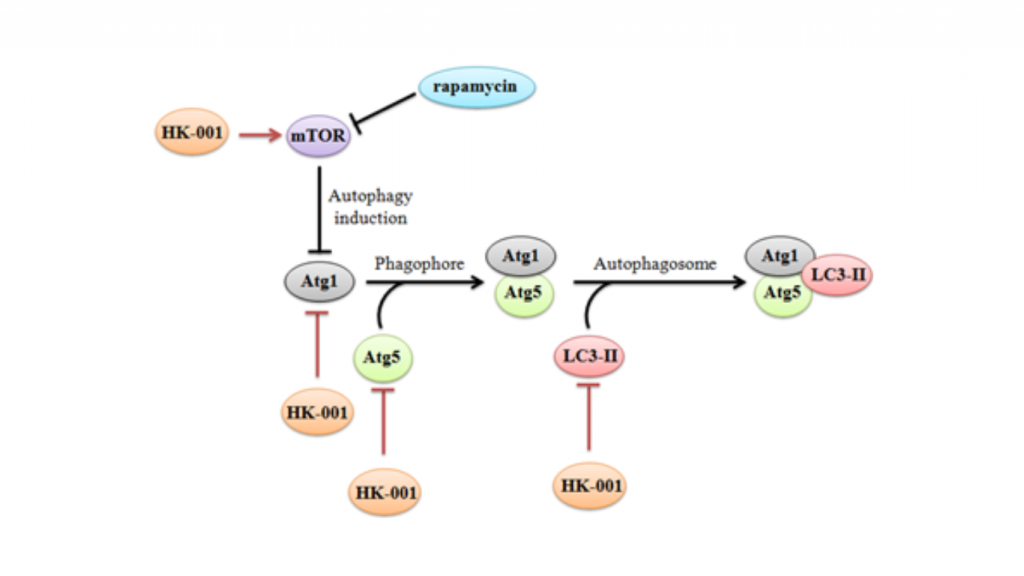
The accumulation of LC3-II is closely related to the role of autophagy, and abnormal accumulation of the LC3-II protein is also considered one of the main causes of ALS deterioration.
In NSC-34-SOD1G93A cell line experiments, it was found that HK-001 can stimulate the activation of mTOR signaling in SOD1G93A mutant cell lines, thereby reducing the accumulation of LC3-II.
Results in animal experiments showed that the accumulation of LC3-II in the spinal nerves of SOD1G93A mice was higher than that in the normal group, and after HK-001 treatment, the mice had higher mTOR activity than the treatment group, and the accumulation of LC3-II was reduced to levels close to the wild type group.
Using specific antibodies for staining motor neurons, it was observed that the number of motor neurons in the HK-001 treatment group was much higher than that in the untreated group, and the number of motor neurons was correlated with BBB scale results. Confirmation was also obtained using LC3-II-GFP transgenic mice, where a decrease in fluorescence intensity was observed in mice treated with HK-001.
These findings confirmed that HK-001 prolongs the lifespan of SOD1G93A mice mainly by regulating the accumulation of LC3-II and downstream autophagy through the phosphorylation/activation of mTOR to achieve the effect of prolonging the lifespan of SOD1G93A mice.
About Riluzole
An orally-administered benzothiazole medication that works by blocking glutamatergic neurotransmission in the CNS. Glutamate itself is an excitatory amino acid that is believed to be associated with the excitotoxicity that causes neurodegeneration in ALS, though the true disease mechanism of ALS is not fully understood, and this is one possible hypothesis.
The drug is not effective in improving survival time or delaying the use of mechanical ventilation for end-stage ALS patients, as indicated on the monograph.
Clinical trial results have shown a median survival of 17.7 months in the treatment group, which is higher than the 14.9 months in the placebo group.
There may be some side effects, such as weakness, nausea, and abnormal liver function indices. About 14% of the subjects discontinued the trial due to these side effects during the clinical trial of this medication.
The neuroprotective mechanism of HK-001
The neuroprotective effect of HK-001 (including the active ingredient EF-API-001) targets calpain and autophagy, both of which are associated with the protein degradation pathway. Inhibition of calpain results in a reduction of abnormal protein degradation. Regulation of autophagy accelerates the clearance of toxic proteins, which is associated with mutant SOD1 accumulation and related neuron degeneration.
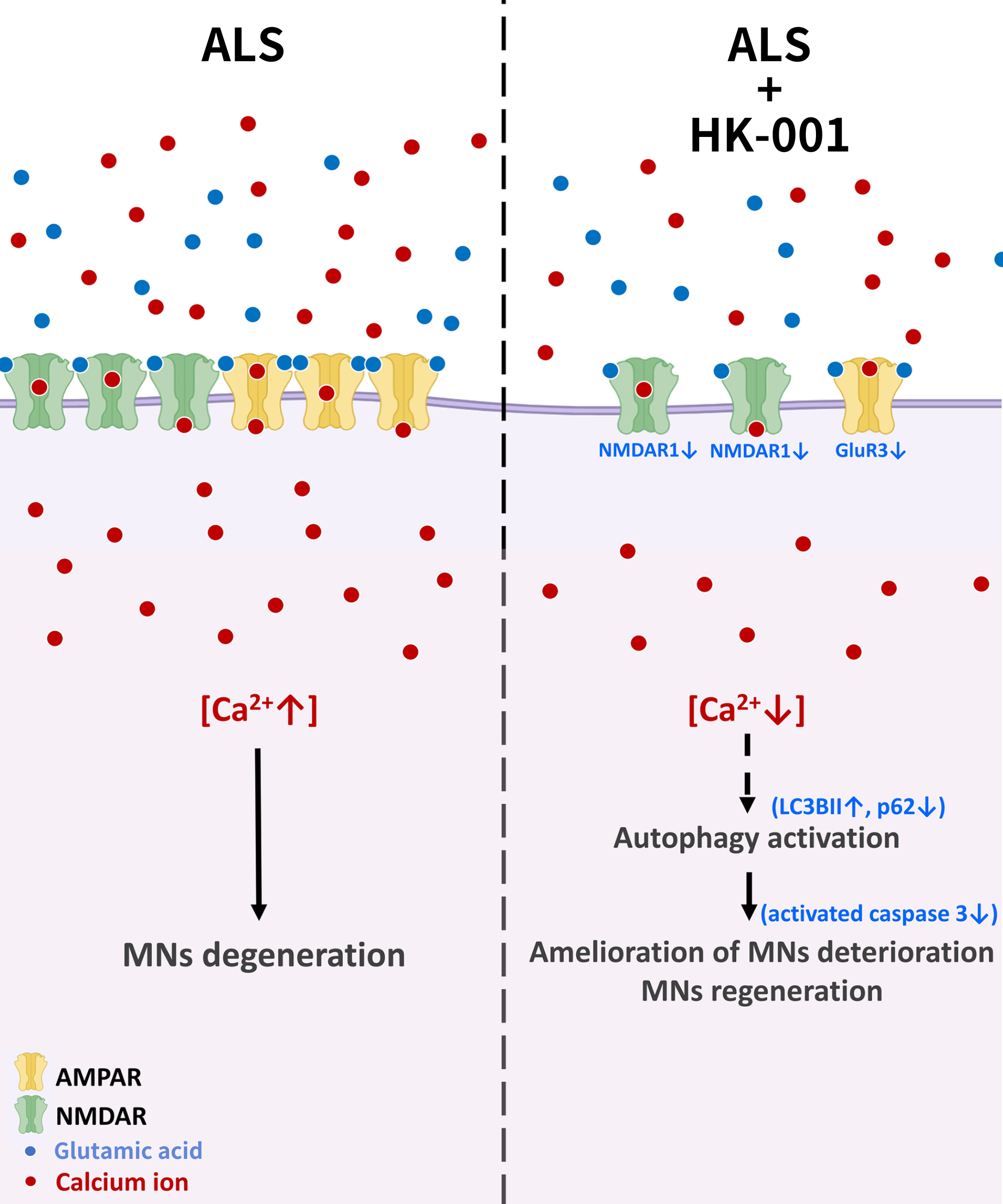
HK-001 ameliorates motor neuron degeneration and promotes regeneration
In ALS, excess glutamic acid release overstimulates calcium-permeable synaptic receptors such as AMPAR (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor) and NMDAR (N-methyl-D-aspartate receptor). Additionally, the reversal of the glutamate transporter increases synaptic glutamic acid and stimulates further calcium influx into postsynaptic neurons. Calcium overload induces cell damage, ultimately leading to apoptosis and neuronal degeneration.
According to our research team’s experimental results, HK-001 can reverse the overexpression of GluR3 (glutamate receptors including glutamate ionotropic receptor AMPA type subunit 3) and NR1, thereby reducing excessive calcium accumulation in postsynaptic motor neurons. Furthermore, HK-001 can activate the autophagic pathway to prevent MN (motor neuron) apoptosis, and prevent or reverse neuron degeneration, reduce the overexpression of glutamate receptor subunits GluR3 and NMDAR1, inhibit excessive glutamate-induced calcium influx, and inhibit hyperactivated apoptosis signaling in motor neurons derived from ALS patient iPSCs harboring the SOD1 G85R mutation.
Full article link: https://doi.org/10.1371/journal.pone.0311573
Awards & Achievements
2014 Received funding support from the A+ Enterprise Innovation R&D Program of the Ministry of Economic Affairs
2017 Awarded 14th National Innovation Award*
2018 IND application approved by USFDA
2019 IND application approved by TFDA
2020 Awarded 16th National Innovation Award – Excelsior Award*
2021 Awarded 18th National Innovation Award – Excelsior Award *
*Taiwan Government Awarded
Patents
Publications
- 2024 | n-Butylidenephthalide recovered calcium homeostasis to ameliorate neurodegeneration of motor neurons derived from amyotrophic lateral sclerosis iPSCs
- 2021 | n-Butylidenephthalide Modulates Autophagy to Ameliorate Neuropathological Progress of Spinocerebellar Ataxia Type 3 through mTOR Pathway
- 2018 | Advances in Patient-Specific Induced Pluripotent Stem Cells Shed Light on Drug Discovery for Amyotrophic Lateral Sclerosis
- 2016 | Autophagic down-regulation in motor neurons remarkably prolongs the survival of ALS mice
Partnering And Collaboration
EFB is looking for the opportunity to cooperate with international pharmaceutical companies or venture capitals.
Please mail us on efbiotech@efbiotech.com