Everfront Biotech Inc.- Rising Star in Pharmaceutical Innovation



EFB'S Mission
Rising Star in Pharmaceutical Innovation
Rising Star in Pharmaceutical Innovation
EFB is committed to fulfilling unmet medical needs by developing orphan drugs to treat the world’s most lethal diseases.
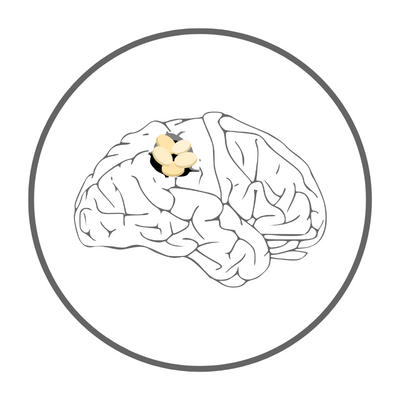
Curing Rare Cancer
PRODUCT HIGHLIGHTS of Cerebraca® Wafer
A Biodegradable Implant Targeting GBM with One-month Sustained Drug Release